Oxidation and Reduction Reactions (Redox) — CBSE Class 10 Chemistry
OIL RIG Made Simple: How Electron Transfer Helps You Truly Understand Redox Reactions
You have seen iron turn orange and flaky after the monsoon. You have watched a cut apple go brown within minutes.
You lit a matchstick this morning. Every single one of these events had something invisible happening underneath — electrons moving from one atom to another. You just did not know its name yet.
The Boy at the Forge
Suresh is fourteen. His father runs a small iron forge near Muzaffarpur, Bihar. Every evening before the rains, Suresh’s job is to coat the iron tools with a thin layer of oil — rods, hammers, chisels, everything. His father told him once, “Oil keeps the air away. Air takes something from iron.” Suresh did not understand what the air could possibly take from a solid lump of metal.
But he followed the rule. And the tools that got the oil stayed shiny. The ones he missed turned rough and orange within three weeks.
Something was moving between the iron and the air. Something invisible. Something that changed the metal permanently once it left.
What Was Actually Leaving the Iron
The iron was not breaking. It was not melting. But it was changing — and that change was real and permanent.
Here is what happens: iron holds electrons. Oxygen in the air pulls those electrons away. Once iron loses those electrons, it becomes a different substance. We call that substance iron oxide. You call it rust.
Now here is the part that surprises most students. When iron loses electrons, oxygen gains them. These two events happen together. You cannot have one without the other. One substance gives. One substance takes. The transfer is the reaction.
That is all a redox reaction is. One substance loses electrons. Another substance is gaining on them. Both are happening at the same moment, in the same place, because of each other.
Now It Gets Its Name
Scientists needed two words for this — one for each half of the transfer.
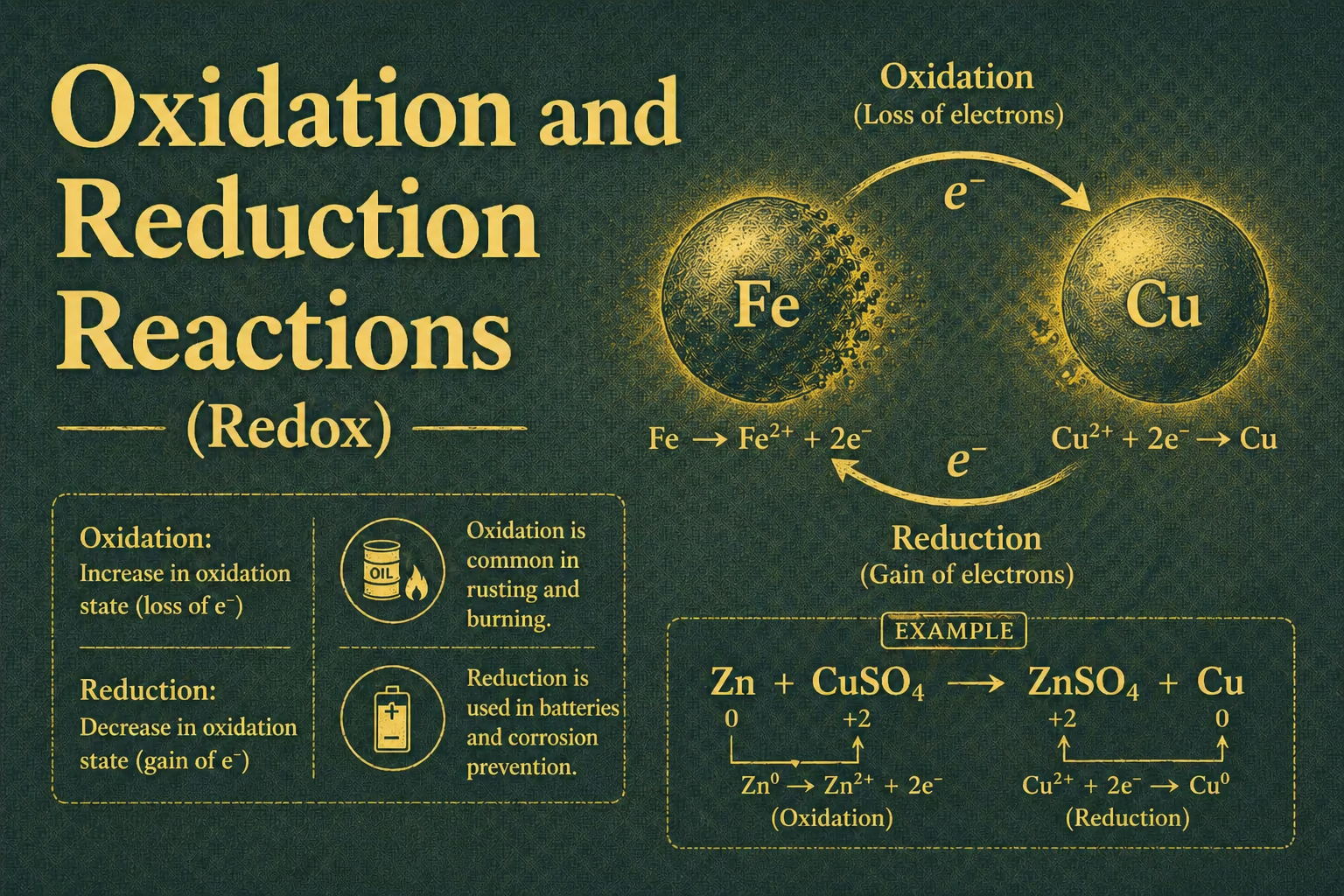
Oxidation is the loss of electrons. When iron loses electrons to oxygen, the iron is being oxidised.
Reduction is the gain of electrons. When oxygen gains those electrons from iron, the oxygen is being reduced.
This is where the memory trick earns its place: OIL RIG.
- Oxidation Is Loss
- Reduction Is Gain
Four words. Carry them into every exam you write from Class 10 onwards.
And one more term: the substance that causes another to lose electrons is called the oxidising agent. The substance that causes another to gain electrons is called the reducing agent. In Suresh’s forge — oxygen was the oxidising agent. Iron was the reducing agent.
How the Transfer Actually Happens — Step by Step
Step 1: Iron atoms at the surface hold electrons loosely. Oxygen atoms in the air have a strong pull on electrons.
Step 2: When iron and oxygen meet, oxygen pulls electrons away from iron atoms. Iron loses electrons. Oxygen gains them. This is the moment of transfer.
Step 3: Iron that has lost electrons is no longer neutral iron. It becomes an iron ion — written Fe²⁺ or Fe³⁺. That “+” sign tells you electrons left.
Step 4: Oxygen that gained electrons combines with iron ions to form iron oxide — Fe₂O₃. That is rust.
Step 5: This is why Suresh’s oil trick worked. Oil blocks oxygen from reaching iron atoms at the surface. No contact. No transfer. No rust.
Every redox reaction follows this same logic — one substance releasing electrons, another pulling them in, a new substance forming at the end.
The Formula — Don’t Panic
This looks like two separate equations. It is. They are showing you the same reaction from two sides at once.
For the rusting of iron:
Oxidation half-reaction (iron loses electrons):
Fe → Fe²⁺ + 2e⁻
Reduction half-reaction (oxygen gains electrons):
O₂ + 4e⁻ → 2O²⁻
The e⁻ symbol means electrons. The arrows show the direction of movement. Iron pushes electrons out. Oxygen pulls electrons in. These two half-reactions add up to the full redox reaction.
You already knew what this meant before you saw the symbols. The formula is just a precise way of writing what Suresh watched happen to his father’s tools every monsoon.
Where You See This Every Day
Burning wood or paper: Carbon in wood loses electrons to oxygen. Carbon is oxidised. Oxygen is reduced. Heat and light are released. This is combustion — a redox reaction you see every time someone lights a fire.
Bleaching a stain: Bleaching powder releases oxygen, which pulls electrons from the coloured molecules in the stain. Those molecules change structure and lose their colour. The stain disappears through electron transfer.
Batteries in your torch: A battery forces electrons to move from one chemical to another through a wire. That electron movement is the electric current. Every battery is a controlled redox reaction.
Cooking with a tawa: When you leave an iron tawa wet, the surface rusts faster. Water speeds up electron transfer between iron and oxygen. Drying the tawa removes the medium that accelerates the reaction.
Where Students Lose Marks
Mistake 1 — Mixing up the agent and the substance. Many students write: “Iron is the oxidising agent because iron gets oxidised.” But look at what the story showed — oxygen does the pulling. Oxygen causes iron to lose electrons. So oxygen is the oxidising agent. The substance being oxidised and the oxidising agent are different things. Always.
Mistake 2 — Treating oxidation and reduction as separate reactions. They are not. They are two halves of one event. Oxidation cannot happen somewhere without reduction happening somewhere else at the same time. If you write about only one half in an exam answer, you have described half a reaction.
Mistake 3 — Thinking “oxidation” always means oxygen is involved. Historically, the word came from oxygen. But today, oxidation means loss of electrons — even when oxygen is nowhere near the reaction. The electron movement is what matters, not the element involved.
The ELIS Ladder — For Every Level
Level 1 — Class 6 to 8 Iron loses electrons to oxygen. That loss is called oxidation. Oxygen gaining those electrons is called reduction. OIL RIG: Oxidation Is Loss, Reduction Is Gain. They always happen together.
Level 2 — Class 9 and 10 (CBSE) A redox reaction involves simultaneous oxidation and reduction. Oxidation = loss of electrons. Reduction = gain of electrons. Oxidising agent = substance that accepts electrons (and causes oxidation in another). Reducing agent = substance that donates electrons (and causes reduction in another).
Half-reactions isolate each part of the transfer:
Oxidation: Fe → Fe²⁺ + 2e⁻
Reduction: O₂ + 4e⁻ → 2O²⁻
Balancing a redox equation means ensuring electrons lost = electrons gained. This is conservation of charge.
Level 3 — Class 11, 12, and Beyond. At higher levels, oxidation state (oxidation number) replaces the electron-counting method for complex reactions. Oxidation state is a bookkeeping tool — an assigned charge that tracks electron distribution across a molecule, even when electrons are not fully transferred (as in covalent bonds). A rise in oxidation number = oxidation. A fall = reduction.
Electrochemistry builds directly on this: in galvanic cells, redox reactions are split physically into two half-cells connected by a salt bridge. The spontaneous electron flow between them generates a measurable voltage. The standard electrode potential (E°) quantifies the tendency of a substance to gain electrons. Students who understand OIL RIG in Class 10 find this extension logical, not alien. Those who memorise without understanding often struggle here.
One important limitation: the simplified OIL RIG model works well for ionic reactions but can mislead in covalent systems where electrons are shared rather than transferred. At the Class 10 level, the model is reliable. In Class 11 and beyond, oxidation state replaces it as the working framework.
In Five Sentences, Three, and One
In five sentences: A redox reaction is a reaction where electrons move from one substance to another. The substance that loses electrons is oxidised. The substance that gains electrons is reduced. These two processes always occur together — you cannot have one without the other. OIL RIG is the memory anchor: Oxidation Is Loss, Reduction Is Gain.
In three sentences: Redox reactions involve the transfer of electrons between substances. Losing electrons is oxidation. Gaining electrons is reduction.
In one sentence: In a redox reaction, one substance gives away electrons while another pulls them in — always at the same time, always inseparably.
Practice Questions
- In the reaction between iron and oxygen, which substance is oxidised and which is reduced? Explain your answer using OIL RIG.
- Magnesium burns in oxygen: 2Mg + O₂ → 2MgO. Identify the oxidising agent and the reducing agent.
- Why can oxidation not occur without reduction happening at the same time?
- A student says: “Oxidation always involves oxygen.” Is this correct? Explain with one example where oxidation occurs without oxygen.
- Write the oxidation half-reaction and the reduction half-reaction for the rusting of iron.
Students Ask These Questions
Q: Is OIL RIG only for Class 10, or does it work later too? OIL RIG is the foundation for everything that follows. In Class 11, you add oxidation numbers. In Class 12, you study electrochemistry. OIL RIG sits underneath all of it. It does not become wrong — it gets extended.
Q: Why is the oxidising agent not the one getting oxidised? This confuses almost everyone at first. The oxidising agent causes oxidation in something else — which means it must accept those electrons itself. So it gets reduced. The naming is about what the agent does to others, not what happens to itself.
Q: Does burning always count as a redox reaction? Yes. Combustion is always a redox reaction. Carbon or hydrogen in the fuel loses electrons to oxygen. Heat and light are the energy released by that electron transfer. Every fire you have ever seen was a redox reaction.
Q: Can a substance be both oxidised and reduced at the same time? Yes — this is called disproportionation. One substance acts as both an oxidising and a reducing agent by transferring electrons to itself. It appears in Class 12. For now, hold the basic model: two different substances, one giving, one taking.
Q: Why does adding oil prevent rusting? Oil creates a physical barrier between iron atoms at the surface and oxygen in the air. Without contact between iron and oxygen, no electron transfer can begin. No transfer means no rust. Suresh’s father was right.
Related reading: Chemical Reactions and Equations — Class 10 | Electrochemistry — Class 12 | Metals and Non-Metals — Class 10 CBSE